Lymphocyte Traffic Patterns
Immunity depends on continuous movement of cells through blood, tissue and lymph (Anderson and Shaw, 1996). Lymphoid cells travel to the secondary lymphoid organs of the spleen, lymph nodes and Peyer's patches to encounter antigens acquired from the environment via blood, lymph or across mucous membranes. Where and by which cells antigens are presented to the trafficking cells has a significant influence on the outcome of the immune response with respect to antibody isotype commitment and future homing preference of memory and effector lymphoid cells.

Lymphocyte traffic patterns, regulated by selective expression of adhesion proteins in peripheral or mucosal lymphatic tissues, permits segregation of immunological memory by causing antigen-primed cells to return to specific anatomic destinations committed to expression of peripheral or mucosal immunity. Among potentially myriad factors, these microenvironments include prevalence of certain cytokines, adhesion to- and costimulation by specific stromal cells, and still unknown tissue factors that favor commitment of B cells to specific immunoglobulin isotypes or T cells to peripheral or mucosal immunity.
Recirculation of a precursor pool of uncommitted lymphocytes from the blood into lymph nodes or mucosal lymphatic tissues and then back to the blood again, integrates immunosurveillance with organ-selective immune functions across the segregated systems. The magnitude of cell traffic reflected by the number of cells returned to the blood in efferent lymph is enormous. Enough lymphocytes recirculate from lymph to blood to replace the total blood lymphocyte pool from 10 to 48 times every 24 hours.
Random and segregated traffic patterns are essential for efficient operation of the two separate but overlapping immune systems in mammalian species. The feat of coordinating an anatomically dispersed immune system (comprised of mobile, circulating, individual and extremely diverse cells) depends upon cell movement and a system of membrane recognition and activation signals. A mixture of integrins, selectins and chemokine receptors expressed by lymphocytes and endothelial cells are involved in precipitating selective emigration of lymphoid subsets from the blood in tissues where specific counter-receptors are displayed on luminal surfaces of endothelial cells. These recognition events could occur in skin, mucosae or specific secondary lymphatic tissues such as Peyer's patches or peripheral lymph node. Receptor ligand interactions allow these cells to find their way around the body, where to adhere to endothelium, when to migrate and how to find where they have to act within tissue.
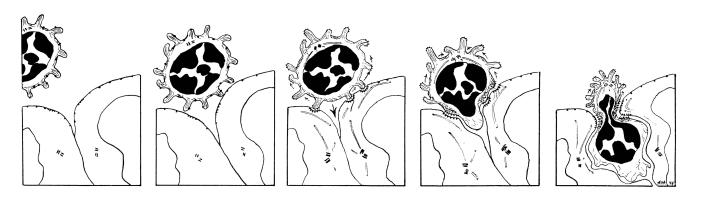
A consensus hypothesis has been offered for explaining the overall process of emigration and to propose ways to explain organ-selective migration as a combinatorial mechanism (Butcher, 1991). Circulating lymphocytes use adhesive selectins to roll on endothelial cell luminal surfaces; they become loosely tethered using selectin-glycam/CD-34 interactions. Upon activation by receptors expressed on the endothelial surface or by chemokines (chemotactic cytokines that emanate from between endothelial cells) the binding characteristics of these receptors are very rapidly (within milliseconds) changed from low affinity to high affinity (Anderson and Shaw, 1993; Ebnet et al., 1996). Different combinations of specific receptor-counter receptor interactions are responsible for each stage and mediate recognition, binding and emigration of cells from the blood. See also: Harvard FLASH VIDEO of cell and molecular events in leukocyte emigration, Ann Ager's article on Lymphocyte Homing, or this nice page from the University of Virginia on the leukocyte emigration sequence of events.
While naive lymphocytes appear to randomly access peripheral lymphoid tissues during recirculation, memory lymphocytes selectively return to the tissues where they first were stimulated by antigen. Precursor lymphocytes (that have not seen antigen in a lymphatic tissue or participated in an immune response) enter all tissues, especially secondary lymphatic tissues, such as Peyers patches, peripheral lymph node or spleen, without any organ selective bias. Shortly after activation by antigen, lymphocytes behave like inflammatory cells and avoid returning to secondary lymphoid tissues, preferring to lodge in skin, gut or inflammatory sites as "acute memory" cells. After maturation occurs, some of these memory cells will follow an organ-selective traffic route determined by the tissue in which that particular lymphocyte encountered the signals to divide differentiate and mature.
When activated by antigen (displayed by an antigen-presenting cell) in any secondary lymphatic tissue, lymphocytes proliferate and undergo profound differentiative changes. They radically change their cell-surface expression of molecules involved in adhesion for a few days after their activation. When these cells emerge from lymphatic tissue as memory cells, their pattern of migration is markedly different. As new memory cells, lymphocytes preferentially home to specific non-lymphoid tissues such as skin or gut (rather than the traffic areas of secondary lymphoid tissues). This behavior fits reasonably well with the increases seen in expression of "inflammatory" adhesion molecules concomitantly with decreases in L-selectin associated with recirculation. Memory CD4 (T-helper) cells express more VLA-4 and LFA-1 and have increased capacity to bind to their ligands VCAM-1 and ICAM-1, whose expression is increased at sites of inflammation.
Expression of particular adhesion proteins and selective secretion of cytokines in peripheral and mucosal lymphoid tissue help to create unique microenvironments (Ebnet et al., 1996). Somehow an influence of this microenvironment is imprinted on the lymphocytes differentiating therein. Differentiation in mucosal lymph nodes results in the induction of integrins that may predispose T cells to interact with vascular addressins expressed on gut venular endothelium and therefore migrate into the gut: included are both the integrins áeß7 for CD8+ IEL cells, which bind to E-cadherin on mucosal epithelial cells (Cepek et al., 1994; Karecla et al., 1995) and á4ß7 for CD4+ lymphocytes, which bind to MADCAM-1 on mucosal venular endothelium and splenic marginal sinus (Hamann et al., 1994; Berlin et al., 1993; Lyons and Parish, 1995; and, Kraal et al., 1995). Conversely, T cells maturing in skin-draining lymph nodes preferentially retain expression of L-selectin (and the sialylated CLA antigen, recognized by HECA 452 monoclonal antibody, which binds to endothelial E-selectin). Both of these integrins appear to facilitate subsequent entry of lymphocytes into the skin or lymph node where L-selectin binds to carbohydrates of the endothelial sialomucins GlyCAM and CD-34.
The leukocyte integrin alpha 4 beta 1 (VLA-4) is activated by a signal emanating from the endothelium and mediates strong binding to VCAM-1 expressed on high endothelial venules (HEV) surfaces. This is thought to facilitate leukocyte emigration between endothelial cells. These differences in the phenotype of emerging T cells must reflect local differences in microenvironment. Such differences have not been defined clearly; however, TGFbeta 1 is believed to be better expressed in mucosal sites and to contribute to the distinctive phenotypes of resident cells. To illustrate, TGF beta 1 upregulates and maintains expression of alpha E beta 7 integrin in T cells destined to enter the intraepithelial compartment where it anchors T cells to E-cadherin on epithelial cells.
B cells, expanding rapidly in primary mucosal lymphoid follicles of very young mammals, undergo diversifying mutations in the genes encoding the antigen-binding sites during VDJ rearrangement (Weinstein et al., 1994). They are then subject to positive selection to generate an antibody repertoire biased toward antigens prevalent in the environment; progeny that do not bind antigen in this milieu undergo apoptosis. Survivors leave the follicles in efferent lymph and enter the blood where they circulate and recirculate as small precursor IgM+/IgD+ B cells. They migrate continuously into all secondary lymphoid tissue until triggered to divide by the appropriate antigen and costimulatory signals.
Functional differentiation of B cells in the circulation depends on whether they are first activated in peripheral lymphoid tissue (lymph nodes and spleen) or in mucosal sites (such as Peyer's patches). They are strongly influenced by prevalent cytokines and unique stromal and accessory cell populations in the respective tissues (Weinstein and Cebra, 1991; Weinstein et al., 1991).
Some precursor B cells are first activated in spleen or lymph node. In that process, they are programmed to be one of the IgG isotypes (rather than IgA). Daughter cells from this clonal expansion leave the lymph node in the efferent lymph, return to the blood and seed the spleen, bone marrow, sites of inflammation and other lymph nodes as plasma cells and "memory" B lymphoblasts.
Other precursor B cells are first activated in Peyer's patches. Those cells become committed to rearrange their immunoglobulin heavy chain genes to express IgA (rather than IgG isotypes noted above); despite having made this commitment, the switch to IgA is often delayed for several days. This cell leaves in the efferent lymph, passes through the mesenteric lymph node (where it may be subject to immunoregulation by T cells it encounters there) and returns to the blood. These cells selectively migrate to the spleen, where furher clonal expansion occurs before the daughter cells are disseminated via the blood to mucosal lamina propria. This process takes 5-7 days, at least four trips in the blood and two to three cell generations (Cebra et al., 1977).

The orderly appearance of recirculating (naive precursor B cells) and non-recirculating (memory or pre-plasma cells) cells in the blood between 2 and 4 days after antigen exposure may provide a valid window for monitoring the status of immune responses before serum antibody can be measured (Anderson and Shaw, 1996). Analysis by flow cytometry may be performed on cells isolated from circulating blood. Two color FACScan analysis will pair red fluorochrome-labeled antigen (which binds to surface immunoglobulins on specific B cells) while green fluorochrome-labeled antibodies can identify whether the circulating cells are committed to peripheral or mucosal immunity or some other phenotypic marker (Quiding-Jarbrink et al., 1995).
It is relatively easy to label recombinant or highly purified antigen for use in identifying and counting antigen-specific B cells. Numerous monoclonal antibodies are available to assist in identifying the functional commitment of cells, including whether the surface immunoglobulins include: IgD (indicating a precursor or primary B cell), or an IgG or IgA isotype (indicating a secondary or memory B cell committed to peripheral or mucosal immunity, respectively). As we become more experienced using flow cytometry, we will be able to estimate the status of an immune response as it evolves. Techniques have even been developed for detecting cytoplasmic proteins by FACScan, including cytokines secreted by T cells that influence B-cell responses (Openshaw et al., 1995; Assenmacher et al., 1994). This is especially important for estimating the potential for mucosal IgA responses, because appearance of surface IgA-positive cells in the blood precedes lodging and differentiation into antibody secretion by several days.
IV. Source References:
1. Anderson, A.O. 1990. "Structure and organization of the lymphatic system." In Immunophysiology. The role of cells and cytokines in immunity and inflammation. Oppenheim, J.J. and E. Shevach (eds.), pp. 14-45, Oxford University Press, NY.
2. Anderson, A.O. and S. Shaw. 1993. T cell adhesion to endothelium: the FRC conduit system and other anatomic and molecular features which facilitate the adhesion cascade in lymph node. Sem. Immunol. 5:271-282.
3. Anderson, A.O., and S. Shaw. 1995. "Lymphocyte Trafficking." Chapter 3, In Clinical Immunology, Principles and Practice. R.R. Rich, T.A. Fleisher, B.D. Schwartz, W.T. Shearer, and W. Strober (eds.), pp 39-49, Mosby-Year Book, Inc., St. Louis, MO.
4. Ebnet, K., E.P. Kaldjian, A.O. Anderson, and S. Shaw. 1995. Orchestrated information transfer underlying leukocyte endothelial interactions. Ann. Rev. Immunol. 14:155-177.
5. Gretz, E.J., E.P. Kaldjian, A.O. Anderson, and S. Shaw. 1996. Sophisticated strategies for information encounter in the lymph node: The reticular network as a conduit of soluble information and a highway for cell traffic. ("Cutting Edge") J. Immunol. 157:495-499.
6. Butcher, E.C. and L.J. Picker. 1996. Lymphocyte Homing and Homeostasis. Science 272:60-66.
7. Gretz, J.E., A.O. Anderson, and S. Shaw. 1997. Cords, channels, corridors and conduits: critical architectural elements facilitating cell interactions in the lymph node cortex. Immunol. Rev. 156:11-24.
8. Anderson, A.O. 1997. New Technologies for Producing Systemic and Mucosal Immunity by Oral Immunization: Immunoprophylaxis in Meals, Ready-to-Eat. In Emerging Technologies for Nutrition Research, National Academy Press, Washington, DC, pp. 451-500.
9. Kuprash, D.V., M.B. Alimzhanov, A.V. Tumanov, A.O. Anderson, K. Pfeffer, and S.A. Nedospasov. 1999. TNF and Lymphotoxin beta Cooperate in the Maintenance of Secondary Lymphoid Tissue Microarchitecture But Not in the Development of Lymph Nodes. J. Immunol 163: 6575-6580.
10. Gretz JE, Norbury CC, Anderson AO, Proudfoot AE, Shaw S. 2000. Lymph-borne chemokines and other low molecular weight molecules reach high endothelial venules via specialized conduits while a functional barrier limits access to the lymphocyte microenvironments in lymph node cortex. J Exp Med 192:1425-1440
11. Kaldjian, E.P., J.E. Gretz, A.O. Anderson, Y. Shi, S. Shaw. 2001. Spatial and molecular organization of lymph node T cell cortex: A labyrinthine cavity bounded by an epithelial sheet of fibroblastic reticular cells anchored to basement membrane-like extracellular matrix. Intl. Immunology 13:1243-1253.
12. Kuprash, D.V., M.B. Alimzhanov, A.V. Tumanov, S.I. Grivennikov, A.N. Shakkov, L.N. Drutskaya, M.W. Marino, R.L. Turetskaya, A.O. Anderson, K. Rajewsky, K. Pfeffer and S. A. Nedospasov. 2002. Redundancy in TNF and LT signaling in vivo: mice with inactivation of the entire TNF/LT locus versus single knockout mice. Mol. Cell. Biol. 22:8626-8634.
13. Anderson, A.O. and S. Shaw. 2005. Conduit for Privileged Communications In the Lymph Node. Preview. Immunity v22 pp3-5. [Author's PDF]
14. Palframan, R.T., Jung, S., Cheng, G., Weninger, W., Luo, Y., Dorf, M., Littman, D.R,, Rollins, B.J., Zweerink, H., Rot, A. and von Andrian, U.H. 2001. Inflammatory Chemokine Transport and Presentation in HEV : A Remote Control Mechanism for Monocyte Recruitment to Lymph Nodes in Inflamed Tissues, J. Exp. Med. V194(9): 1361-1374.
15. Baekkevold, E.S., Yamanaka, T., Palframan, R.T., Carlsen, H.S., Reinholt, F.P., von Andrian, U.H., Brandtzaeg, P., and Haraldsen, G. 2001. The CCR7 Ligand ELC (CCL19) Is Transcytosed in High Endothelial Venules and Mediates T Cell Recruitment. J. Exp. Med., v193(9):1105-1112.
16. Penna, G., Sozzani, S. and Adorini, L. 2001. Cutting Edge: Selective Usage of Chemokine Receptors by Plasmacytoid Dendritic Cells. J. Immunol. 167: 1862-1866.
17. Ingulli, E, DR Ulman, MM Lucido, and Marc K Jenkins. 2002. In Situ Analysis Reveals Physical Interactions Between CD11b+ Dendtritic Cells and Antigen-Specific CD4 T Cells After Subcutaneous Injection of Antigen. J. Immunol. 169: 2247-2252.
18. Ohtani, O, Ohtani, Y, Carati, CJ, and Gannon, BJ. 2003. Fluid and Cellular Pathways of Rat Lymph Nodes in Relation to Lymphatic Labyrinths and Aquaporin-1 Expression. Arch. Histol. Cytol. 66(3):261-273.
19. Nolte, MA, JAM Belien, I Schadee-Eestermans, W. Jansen, WWJ Unger, N van Rooijen, G Kraal and RE Mebius. 2003. A Conduit System Distributes Chemokines and Small Blood-borne Molecules through the Splenic White Pulp. J. Exp. Med 198:505-512
20. Bejenoff, M, S Granjeaud and S Guerder. 2003. The Strategy of T Cell Antigen-presenting Cell Encounter in Antigen-draining Lymph Nodes Revealed by Imaging of Initial T Cell Activation. J. Exp. Med. 198:715-724
21. Itano AA, SJ McSorley, RL Reinhardt, BD Ehst, E Ingulli, AY, Rudentsky and Marc K Jenkins. 2003. Distinct Dendritic Cell Populations Sequentially Present Antigen to CD4 T Cells and Stimulate Different Aspects of Cell-Mediated Immunity. Immunity 19: 47-57.
22. von Andrian, UH and T Mempel. 2003. Homing and Cellular Traffic in Lymph Nodes. Nature Reviews Immunology 3: 867-878. [PubMed]
23. Thomazy VA, Vega F, Medeiros LJ, Davies PJ and Jones D. 2003. Phenotypic modulation of the stromal reticular network in normal and neoplastic lymph nodes: tissue transglutaminase reveals coordinate regulation of multiple cell types. Am J Pathol. 63: 165-74.
24. Mempel, TR, SE Henrickson, and UH von Andrian. 2004. T cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427: 154-159. [PubMed]